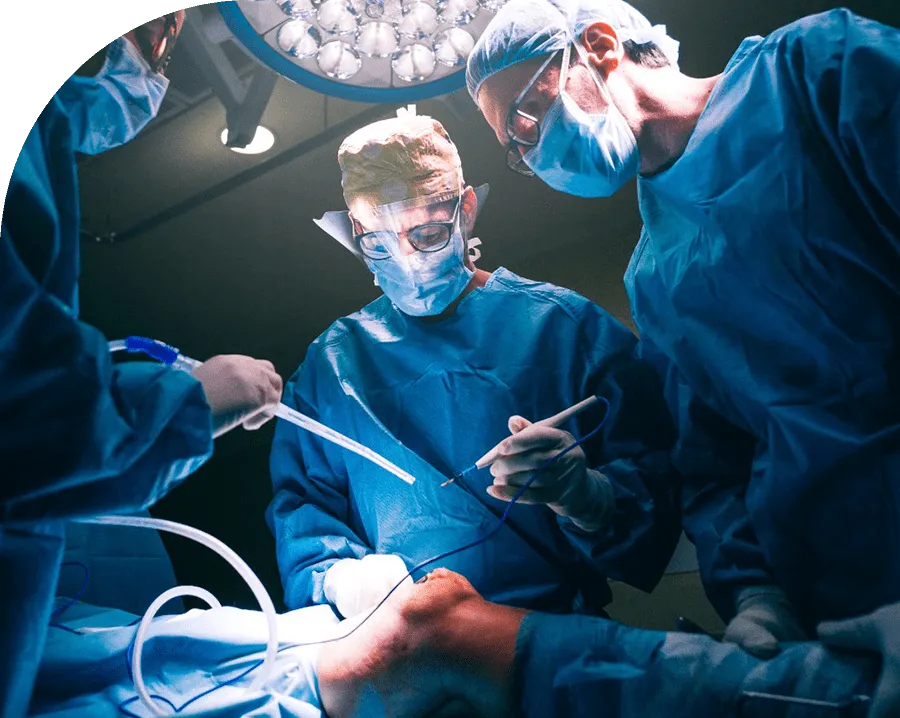
Guided implant surgery
Implant site preparation under digital 3D guidance with magnetodynamic osteotomes. Sleeve-compatible workflow, no rotary friction heat under the surgical guide.
Magnetodynamic surgical technology · 13 years worldwide, 10+ years in the U.S.
Established U.S. clinical infrastructure · 30+ countries on five continents · Peer-reviewed evidence base
OSSEOTOUCH develops and distributes the Magnetic Mallet — a magnetodynamic device for oral and implant surgery, adopted by clinicians worldwide for over 13 years and serving U.S. clinicians for over a decade. Bone-preserving site preparation without rotary friction heat.

OSSEOTOUCH develops and distributes the Magnetic Mallet — a magnetodynamic system for oral and implant surgery. The technology has been adopted by clinicians worldwide for over 13 years and has served the U.S. dental market for over a decade.
The handpiece generates a calibrated magnetic impulse that transfers controlled axial force to interchangeable surgical instruments — without rotary cutting and without rotary friction heat in the magnetodynamic phase. Bone is laterally displaced rather than removed: vital bone displacement.
The mission is straightforward: make oral surgery simpler for the clinician and more comfortable for the patient. No vibration, no friction heat, no rotary noise — just calibrated force, applied with millimetric precision.
Today the Magnetic Mallet is in active clinical use across more than 30 countries on five continents, supported by certified clinical and academic partners.
Three numbers that frame the system: how long the technology has been clinically adopted, on which markets, and how broad the global footprint is today.
The principle is straightforward: minimize impact time, maximize controlled force. The result is a calibrated impulse that displaces and compacts bone rather than removing it.
Clinical questions, configuration, Try Before You Buy (when available), financing — handled by an OSSEOTOUCH specialist in a single dedicated session.
The Magnetic Mallet is a modular system: one device, multiple dedicated kits. Each application uses the same magnetodynamic principle to deliver predictable, bone-preserving outcomes.
Implant site preparation under digital 3D guidance with magnetodynamic osteotomes. Sleeve-compatible workflow, no rotary friction heat under the surgical guide.
Crestal-approach sinus elevation for both small and larger lifts. Controlled, no irrigation in the magnetodynamic phase. Designed to support membrane integrity.
Progressive split-crest expansion with sequential inserts. Addresses severe horizontal atrophy without grafting in selected cases — subject to clinical judgment.
Progressive wedge effect within the periodontal space. Designed to help preserve alveolar bone, with reduced trauma compared to conventional elevators and luxators.
Non-rotary cancellous condensation with dedicated tips. Cold procedure, no irrigation in the magnetodynamic phase, even in selected challenging scenarios.
Complete alternative to rotary preparation. No rotary friction heat in the magnetodynamic phase. Supports primary stability through cancellous condensation.
Longitudinal magnetodynamic traction designed to reduce stress on the underlying abutment when clinical conditions allow. Particularly useful on zirconia.
The magnetodynamic system also extends, in early development, to adjacent surgical fields where precision and controlled force matter.

Surgical procedures on companion and farm animals using the same minimally invasive technology. Designed to reduce intra-operative stress and recovery times.
Precise management of nasal bone structures with controlled force. Supports predictability of aesthetic and functional outcomes.
Real registration data from current certificates. No marketing claims — facts that can be verified.
A surgical technology that generates calibrated magnetic impulses to perform minimally invasive oral surgery. The handpiece transfers controlled axial force to interchangeable surgical instruments without rotary cutting and without rotary friction heat in the magnetodynamic phase. Bone is laterally displaced (vital bone displacement) rather than removed.
Magnetodynamic surgical technology has been adopted by clinicians worldwide for over 13 years and has served the U.S. dental market for over a decade.
Registered with the U.S. FDA under Establishment Registration 3011922183 and product codes EIS · KDG · GEY (21 CFR 872.4565 / 878.4820). Manufacturer: Meta Ergonomica S.r.l. (Italy). U.S. Agent: Thema USA, New York. ISO 13485:2016 quality management system (Certificate ICIM-13485-050738-00). CE-marked.
Atraumatic extraction, crestal sinus lift, split crest (horizontal ridge expansion), implant site preparation, guided implant surgery, atraumatic crown and bridge removal, GBR pin fixation, and All-on-X workflows.
No. The magnetodynamic system also extends to veterinary surgery and rhinoplasty, with dedicated solutions in early development.
OSSEOTOUCH operates in the United States through its U.S. commercial team, supporting clinicians with onboarding, technical and clinical questions, financing, and the Try Before You Buy program (when available). U.S. Agent for FDA listing: Thema USA, New York.
Pick a date and time. We will contact you.
cs@osseotouch.com
Direct chat with our team.
OSSEOTOUCH devices are medical devices for professional use, restricted to qualified dental professionals according to the instructions for use. United States: registered with the U.S. FDA under Establishment Registration 3011922183 and product codes EIS · KDG · GEY (21 CFR 872.4565 / 878.4820). U.S. Agent: Thema USA, New York. ISO 13485:2016 (Certificate ICIM-13485-050738-00 / IQNET IT-149738). CE-marked. Manufactured by Meta Ergonomica S.r.l. (Italy). Clinical outcomes depend on case selection, surgical technique, and protocol; the information on this page is for educational purposes and does not replace the clinical judgment of the professional.