Magnetic Mallet
Surgical unit for all procedures
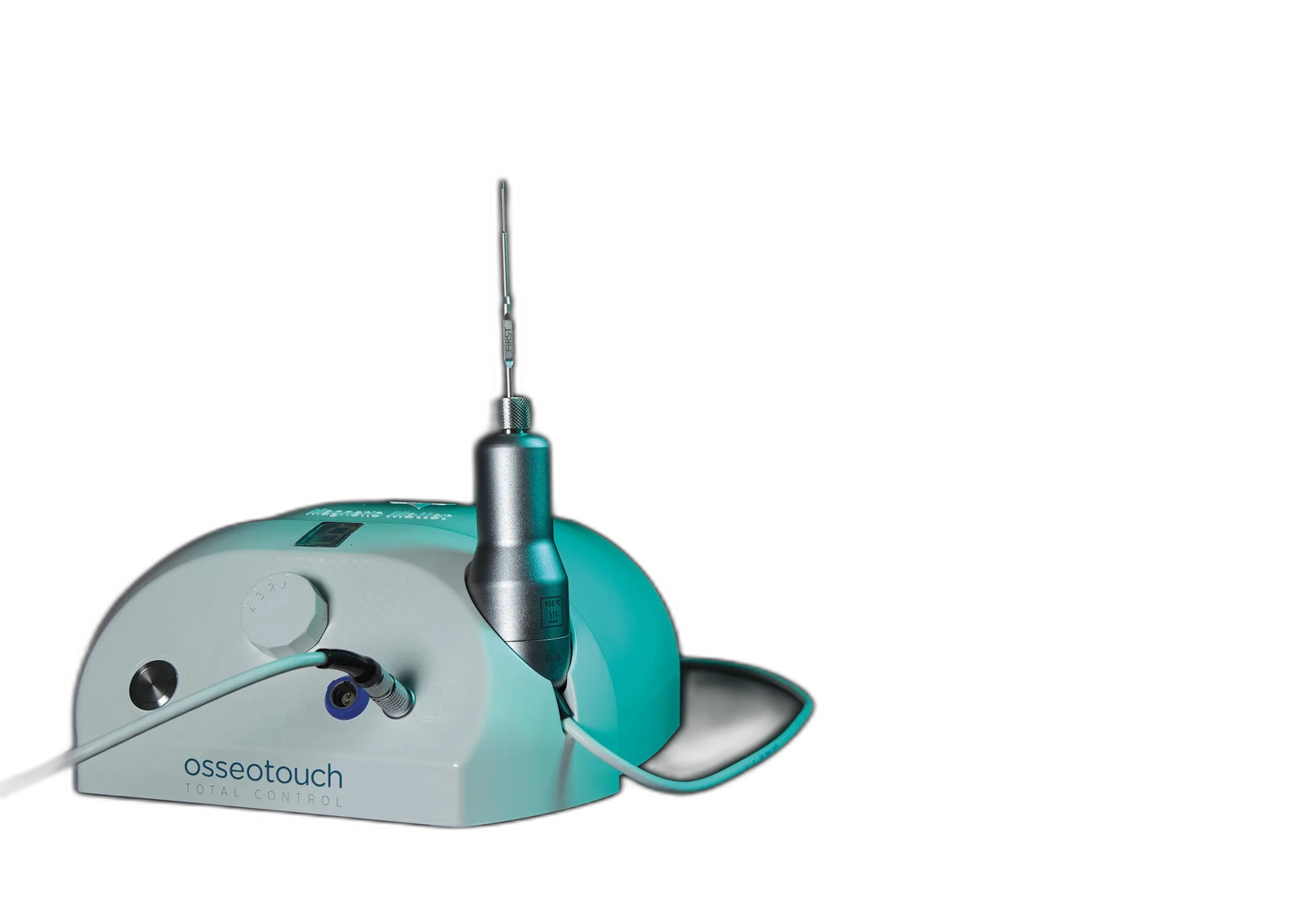
-
Catalog (USA-ENG)
Full product overview of the Magnetic Mallet system: device, Plus Handpiece, foot pedal, and the complete osteotome line for the U.S. market.
Download PDF -
User Manual (multilingual)
Instructions for use of the Magnetic Mallet medical device. Multilingual edition (ITA, ESP, POR). The English-only manual is being prepared for the U.S. release.
Download PDF
Easyroot
Surgical unit for atraumatic extraction

Surgical Instruments
Osteotomes and instruments for Magnetic Mallet & Easyroot

Crown Remover (Levacorone)
Magnetic Mallet instrument for crown removal
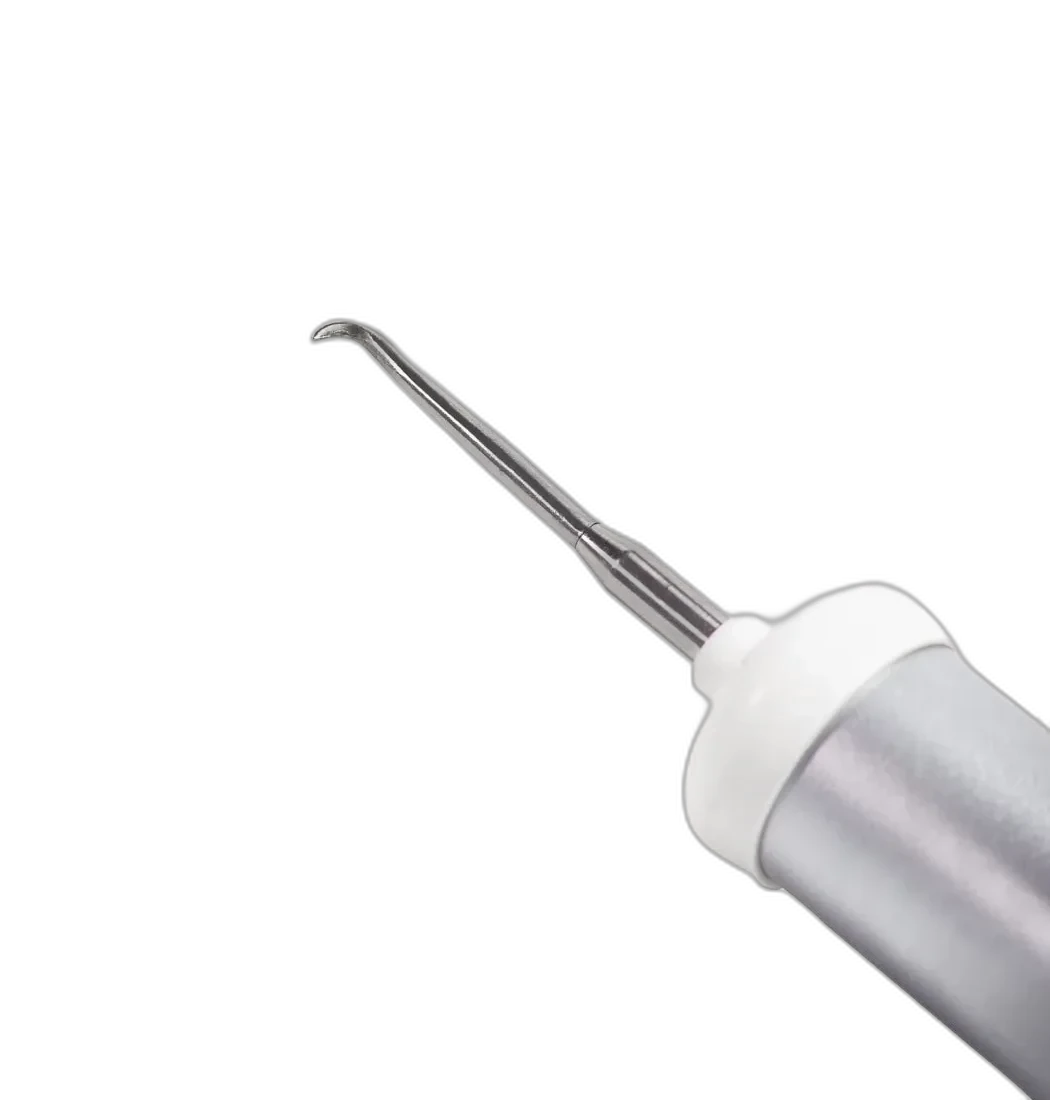
Conex-K & Only One Implants
OSSEOTOUCH implant line

-
Implant Catalog
Complete range of the Conex-K and Only One implant systems with technical and dimensional specifications.
Download PDF -
Surgical Protocol
Operative sequence for the placement of Conex-K and Only One implants.
Download PDF -
Prosthetic Components
Catalog of the prosthetic componentry compatible with the Conex-K and Only One implant lines.
Download PDF
Certifications
Meta Ergonomica S.r.l. — manufacturer of the Magnetic Mallet and OSSEOTOUCH device line — operates a quality management system for the design, manufacturing, distribution and servicing of medical devices.
- ISO
ISO 13485:2016 Certificate
Quality management system for the design, manufacturing, distribution and servicing of medical devices. Certificate ICIM-13485-050738-00 issued to Meta Ergonomica S.r.l.
Download PDF - CE
CE MDR 2017/745 Certificate
CE certificate issued by the European Notified Body in compliance with Medical Device Regulation (EU) 2017/745 on medical devices.
Download PDF - FDA
FDA Registration — Magnetic Mallet
U.S. Food and Drug Administration registration certificate for the Magnetic Mallet device under product code EIS (21 CFR 872.4565).
Download PDF - FDA
FDA Registration — Easyroot
U.S. Food and Drug Administration registration certificate for the Easyroot device.
Download PDF
Need a document that's not here?
Email or message the OSSEOTOUCH team — we reply within one business day.
