One-handed insertion
The pin applicator threads onto the Magnetic Mallet handpiece. One hand delivers the pin; the other stays free to position and tension the membrane — no second instrument, no second pair of hands.
Membrane fixation in a single tap
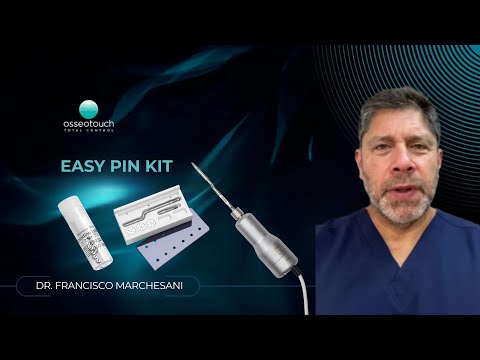
GBR Pin Insertion System · titanium pins for membrane fixation, one-hand workflow with the Magnetic Mallet
Titanium pins for barrier membrane fixation in guided bone regeneration. The applicator threads onto the Magnetic Mallet handpiece and delivers the pin in a single calibrated impulse — one-handed, no surgical hammer. Two pin lengths: 3.1 mm (XL) and 3.5 mm (XXL), sterile, single-use.

Traditional pin insertion demands two hands and two tools — surgical hammer plus manual punch. Force is variable and operator-dependent: tap too hard and the pin bends; too soft and it sits proud. Posterior and lingual sites make it worse, often forcing an assistant to hold the membrane while both surgeon's hands are occupied.
Easy-Pin threads onto the Magnetic Mallet handpiece and delivers the pin in a single 50-microsecond impulse — calibrated, operator-independent. One hand on the handpiece, the other free to position the membrane. Designed to reduce pin bending, support more consistent seating, and access lingual and posterior sites that hammer-and-punch protocols can't easily reach.
One handpiece. Two pin lengths. Six reasons fixation becomes faster, more predictable, and less hand-intensive in every GBR procedure.
The pin applicator threads onto the Magnetic Mallet handpiece. One hand delivers the pin; the other stays free to position and tension the membrane — no second instrument, no second pair of hands.
The 50-microsecond magnetodynamic impulse drives the pin perpendicular to bone in a single shot. The system is designed to reduce the glancing strikes and progressive bending typical of manual hammer-and-punch protocols.
Force is delivered by the device, not the surgeon's wrist. The same insertion energy at 8 a.m. and 6 p.m., across the first pin and the twelfth — operator-independent and fatigue-resistant.
The angled applicator and the compact handpiece reach lingual, palatal, and distal sites where a hammer-and-punch protocol struggles. Membranes get fixed where they actually need fixing.
3.1 mm (XL) and 3.5 mm (XXL) titanium pins, packaged five per pack, sterile, single-use. Pick the length that matches cortical thickness and bone density.
Solo workflow: the surgeon places the membrane and fixes it without needing an assistant to hold tension. Frees up the team and shortens the chair time on long GBR cases.
Talk with our specialist to evaluate Easy-Pin for your GBR workflow, check compatibility with your existing Magnetic Mallet handpiece, or request a demo on a clinical case mix.
The base kit contains the magnetodynamic applicator and an autoclavable storage box. Titanium pins are sold separately in two lengths — sterile, single-use, five per pack.

The autoclavable surgical box hosts the INSEP applicator — which threads onto the Magnetic Mallet handpiece and integrates the PNTEP tip for pin engagement and release — plus dedicated pin holders. Reusable case after case with standard autoclave sterilization.
| Code | Length | Pack | Indication |
|---|---|---|---|
| PIN-XL | 3.1 mm | 5 pins | Standard cortical thickness (posterior maxilla, mandible) |
| PIN-XXL | 3.5 mm | 5 pins | Thicker cortex or higher membrane tension |
Biocompatible surgical titanium, sterile single-use foil pouches. Power level 1–2 of 4 for typical cortical bone, no pre-drilling required.
Available now Order Easy-Pin directly through OSSEOTOUCH USA — fast nationwide delivery, dedicated clinical support.
Buy onlineEasy-Pin handles every barrier-membrane fixation scenario in the regenerative workflow — from straightforward GBR to complex combined cases — with the same magnetodynamic insertion principle and the same one-handed technique.
Stabilize barrier membranes (resorbable or non-resorbable) over the bone defect. The titanium pin anchors the membrane to the underlying cortical bone with a single calibrated impulse — supporting controlled membrane fixation and predictable insertion depth.
When particulate bone graft is placed under a barrier membrane, pin fixation prevents micromovement during the early healing phase. Easy-Pin positions the membrane firmly over the graft volume without disturbing the particulate material.
The angled applicator and the maneuverable Magnetic Mallet handpiece reach lingual, palatal, and distal sites where a hammer-and-punch protocol is mechanically restricted. Including the posterior mandible.
When implant placement and GBR are performed in the same session, the workflow benefits from one-handed pin insertion: the surgeon retains control of the membrane and surrounding tissue without an assistant actively holding instruments.
Order now Add Easy-Pin to your GBR workflow — base kit + initial titanium pin packs through OSSEOTOUCH USA with fast nationwide delivery.
Buy onlineTwo documented clinical cases — Easy-Pin used for barrier membrane fixation during real GBR procedures, by international clinicians who use it in their daily workflow.

Easy-Pin is a magnetodynamic system for inserting titanium pins to fix barrier membranes during guided bone regeneration (GBR). The pin applicator threads onto the OSSEOTOUCH Magnetic Mallet handpiece and delivers the pin in a single calibrated 50-microsecond impulse. Used for GBR membrane fixation, particulate bone graft stabilization, hard-to-reach lingual and posterior sites, and combined implant-plus-GBR procedures.
Manual protocols require two hands and two instruments — a surgical hammer and a manual punch. Force is variable and operator-dependent, with risk of pin bending and inconsistent insertion depth. Easy-Pin delivers a calibrated 50-microsecond impulse with a single one-handed motion: the pin enters perpendicular to bone, designed to reduce pin bending and support more consistent seating.
Not for the pin insertion step. With Easy-Pin the surgeon operates the handpiece with one hand and keeps the other hand free to position and tension the membrane. Solo workflow on the fixation step shortens chair time and removes a coordination dependency.
Two lengths: 3.1 mm (XL) and 3.5 mm (XXL). Both are biocompatible titanium, sterile single-use, packaged five per pack. Pins are sold separately from the base kit. Choose XL for typical cortical thickness, XXL for thicker cortex or higher membrane tension.
Yes. Titanium pins fix any barrier membrane commonly used in GBR — resorbable collagen membranes, non-resorbable PTFE/d-PTFE/titanium-mesh-reinforced membranes — to the underlying cortical bone. The pin acts as a mechanical anchor without restricting membrane material choice.
Yes. The applicator's angled geometry combined with the maneuverable Magnetic Mallet handpiece reaches lingual, palatal, and distal-mandibular sites where conventional manual hammer-and-punch protocols are mechanically restricted. Particularly valuable for posterior mandibular GBR.
Power level 1–2 of 4 is typical for cortical bone in standard GBR sites. Higher levels are reserved for unusually dense cortical bone. The 50-microsecond impulse is below the threshold typically perceived as vibration.
The OSSEOTOUCH Magnetic Mallet is registered with the U.S. FDA under Establishment Registration 3011922183 and product codes EIS · KDG · GEY (21 CFR 872.4565 / 878.4820). U.S. Agent: Thema USA, New York. The Easy-Pin applicator fits new-generation Magnetic Mallet handpieces and is fully autoclavable.
Whether you're moving from manual hammer-and-punch protocols or adding GBR pin fixation to your existing Magnetic Mallet workflow, our team will help you choose the configuration and run a demo at your office or via video call.
Pick a date and time. We will contact you to discuss your workflow.
cs@osseotouch.com
Direct chat with our team.
U.S. ordersOrder Easy-Pin through OSSEOTOUCH USA — fast nationwide delivery and clinical support.
Easy-Pin is part of the OSSEOTOUCH Magnetic Mallet system, manufactured by Meta Ergonomica S.r.l. (Italy). United States: registered with the U.S. FDA under Establishment Registration 3011922183 and product codes EIS · KDG · GEY (21 CFR 872.4565 / 878.4820). U.S. Agent: Thema USA, New York. ISO 13485. Use restricted to qualified dental professionals according to the instructions for use.