Predictable
Calibrated magnetic impulse — same force on every activation, not surgeon-dependent. Four force levels selected on the console. The third case of the day is repeated with the same parameters as the first.
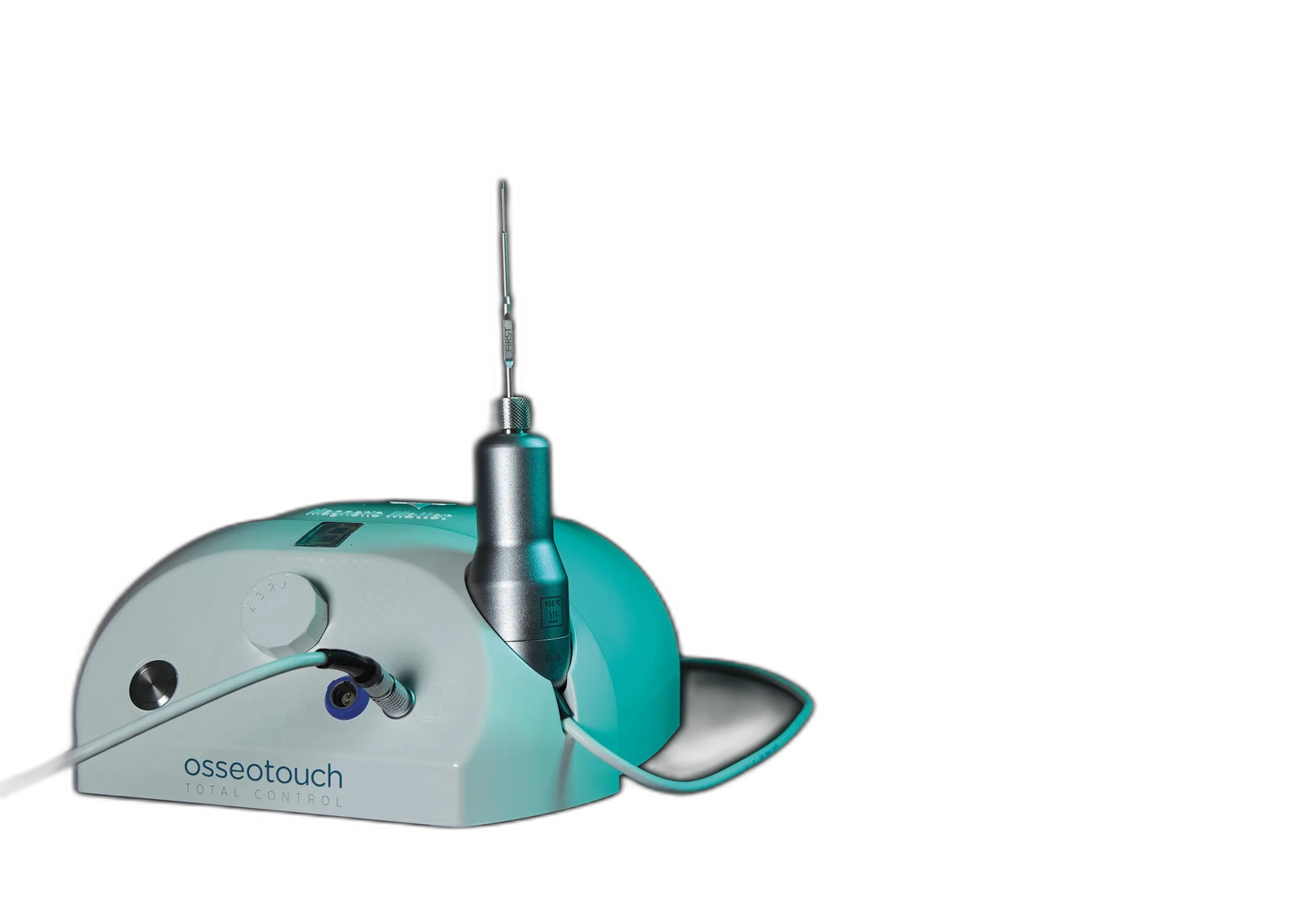
Non-rotary magnetodynamic surgery · bone-preserving site preparation
10+ years on the U.S. market · Calibrated 80-microsecond impulse · 4 force levels · 1 mm per impulse
A calibrated magnetic impulse transfers controlled axial force to interchangeable surgical instruments — without rotary cutting and without rotary friction heat in the magnetodynamic phase. Bone is laterally displaced, not removed. Predictable, controllable, repeatable osteotomies.
Conventional rotary drilling excavates bone, generates frictional heat at the bone-bur interface, and requires irrigation that can wash away osteogenic factors. In low-density (D3–D4) maxillary bone, every micron removed is a micron lost — and the trajectory is committed the moment the cortical entry is taken.
A calibrated 80-microsecond magnetic impulse transfers controlled axial force. Bone is laterally displaced rather than excavated — the matrix at the surgical site is conserved. No rotation, no rotary friction heat in the magnetodynamic phase. The trajectory can typically be corrected mid-osteotomy.
The clinical case for the Magnetic Mallet rests on three pillars — not on a single feature.
Calibrated magnetic impulse — same force on every activation, not surgeon-dependent. Four force levels selected on the console. The third case of the day is repeated with the same parameters as the first.
1 mm advancement per impulse, foot-pedal driven. The surgeon controls direction, intensity, and depth. The trajectory can typically be corrected mid-osteotomy — control not available with rotary subtraction.
Non-rotary preparation. No rotary cutting, no rotary friction heat in the magnetodynamic phase. Bennardo 2022 systematic review: zero BPPV cases vs. 7 with manual mallet across 525 patients reviewed.
U.S. orders · available now
Bring the Magnetic Mallet to your practice — order through OSSEOTOUCH USA, fast nationwide delivery and dedicated clinical support.
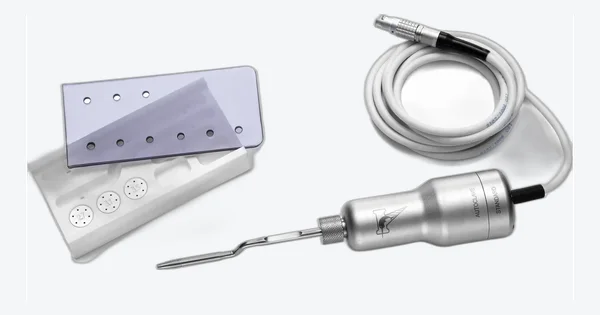
Interchangeable instrument families thread onto the handpiece, designed for each procedure. The Magnetic Mallet is the device; the kits define the workflow.

Crestal sinus lift

Non-rotary osseodensification

Pterygoid implants

Rotary bur-free site prep

Atraumatic extractions

Guided implant surgery

Premium complex extractions
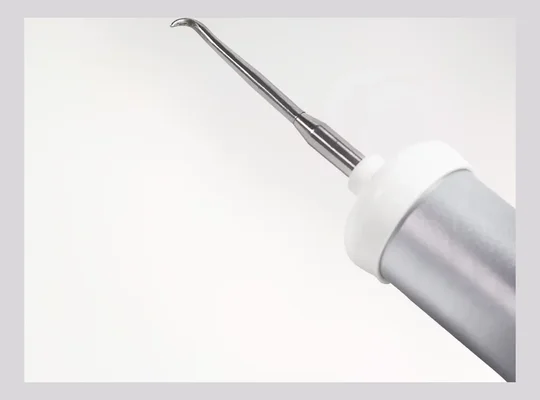
Membrane fixation
Crown and bridge removal

Split crest

Lancet osteotome

20° mandibular access
Atraumatic extraction, sinus lift, split crest, implant site preparation, guided surgery, crown removal, All-on-X. Non-rotary, with interchangeable instrument families.
Targeted luxation designed to help preserve alveolar bone. Conserved geometry supports immediate-load planning when clinically appropriate.
Sinus elevation via crestal approach with vital bone displacement. In selected cases, may be performed without graft material — subject to clinical judgment.
Horizontal expansion of atrophic ridges with same-session implant placement.
Osteotomy without bone removal. Trajectory can typically be adjusted mid-procedure — control not available with rotary subtraction.
Osteotomy without rotary burs. Supports torque values consistent with immediate-load protocols, subject to clinical judgment.
Atraumatic removal in 1-2 impulses, designed to reduce stress on the underlying abutment when clinical conditions allow.
Clinical questions, configuration, Try Before You Buy (when available), financing — handled by an OSSEOTOUCH specialist in a single dedicated session.
Three published configurations to start without choosing kit by kit. Each remains expandable at any time.
Device + Essential Extraction Kit + Silver Osteotome Kit. 10 instruments. Atraumatic extractions, implant site preparation, free-hand crestal sinus lift.
Discover Infinity → PREMIUM STANDARDDevice + Essential + Black Ruby + Kit Elevate. 14 instruments. Next-generation rotary bur-free site preparation and millimetric-stop sinus lift. DLC coating.
Discover Total Combo → FLAGSHIPDevice + Essential + BLEXO + Levacorone + Black Ruby + Kit Elevate. 26 instruments. The most complete published configuration.
Discover Master →
U.S. orders · ships nationwide
Order the Magnetic Mallet through OSSEOTOUCH USA — fast nationwide delivery, dedicated clinical support.
Supported by peer-reviewed publications across in-vitro, animal, and human-clinical study designs. Study type is reported alongside each citation.
Materials (MDPI) · pilot animal study
Biomolecular, histological, clinical and radiological analyses on MM-prepared dental implant bone sites. Significantly higher new bone formation, osteoblast count, and BMP-4 expression vs. rotary controls in the animal model.
DOI →Exploration of Medicine · in-vitro porcine rib
In-vitro comparative study on three parameters — accuracy, bone loss, and temperature. All three significantly better with Magnetic Mallet (P < 0.01) in the porcine rib model. Not in-vivo, not clinical on humans.
DOI →J. Personalized Medicine (MDPI) · systematic review
Systematic review of 14 studies covering 619 extractions and 880 implants. Zero BPPV cases in the magnetodynamic group vs. 7 in the manual-mallet control across the reviewed cohort.
DOI →Transparency note: several authors of the cited studies are affiliated with the University of Genoa and the University of Turin and have collaborated with Meta Ergonomica S.r.l. on the development of the technology. Cited studies include pilot animal study (Schierano 2021), in-vitro porcine rib model (Baldi 2024), and systematic review (Bennardo 2022) — they are not equivalent to large randomized controlled trials on humans.
The handpiece generates a calibrated magnetic impulse that transfers controlled axial force to the surgical instrument. Bone behaves as an elastic structure under axial load — laterally compacted, the matrix preserved.
Each approach is evidence-based and has its place. The technical principle — and the resulting intra-operative experience — differ.
Rotary drills excavate bone, generate frictional heat, require irrigation. The Magnetic Mallet displaces and compacts bone laterally without rotation — preserves native bone matrix, avoids rotary friction heat, keeps the field dry during the magnetodynamic phase.
Manual mallets are operator-dependent: impulse intensity varies with hand strength, fatigue, chair position. Bennardo 2022 reports 7 BPPV cases with manual mallet vs. zero with Magnetic Mallet across 525 patients in the reviewed cohort. Calibrated impulse removes operator variability.
A magnetodynamic medical device for oral and implant surgery. Listed under Establishment Registration 3011922183 and product codes EIS · KDG · GEY (21 CFR 872.4565 / 878.4820). The handpiece generates a calibrated 80-microsecond magnetic impulse — non-rotary site preparation. ISO 13485. CE-marked. Used by clinicians in the United States for over a decade.
Calibrated magnetic impulse. Four force levels (75 / 90 / 130 / 260 daN), 1 mm advancement per impulse, 80-microsecond impact duration. Foot-pedal control, single-hand pen-style grip. Bone is laterally displaced rather than removed. No rotation, no rotary friction heat in the magnetodynamic phase.
Registered with the U.S. FDA under Establishment Registration 3011922183 and product codes EIS · KDG · GEY (21 CFR 872.4565 / 878.4820). Manufacturer: Meta Ergonomica S.r.l. (Italy). U.S. Agent: Thema USA, New York. ISO 13485. CE-marked. Over a decade on the U.S. market.
No appreciable heat in the magnetodynamic phase. The 80-microsecond impulse generates negligible heat at the bone-instrument interface, reported significantly lower than rotary drilling in selected in-vitro studies (P < 0.01, Baldi 2024 — porcine rib model).
It supports densification of low-density (D3-D4) cancellous bone, documented radiographically (CBCT) in selected studies. Schierano 2021 (pilot animal study) reports BMP-4 expression up to 6× higher and significantly more osteoblasts in MM-prepared sites compared to rotary burs in the animal model.
Mixed designs. Bennardo 2022 systematic review aggregates 14 studies including human clinical data (619 extractions / 880 implants). Schierano 2021 is a pilot animal study. Baldi 2024 is an in-vitro porcine rib study. The body of evidence is peer-reviewed across in-vitro, animal, and human-clinical study designs.
Yes — for extractions, split crest, apical root removal in proximity of the inferior alveolar nerve, and All-on-X angled accesses. In dense D1 mandibular bone, conventional rotary drilling typically remains the efficient choice for the principal preparation.
Over a decade. According to company records, clinicians in the United States have worked with the Magnetic Mallet system for years.
Pen-style grip, single hand. Protocols are similar to rotary protocols. Hands-on training is recommended before clinical use; OSSEOTOUCH includes structured onboarding and remote clinical support for the first case (when available).
Talk with the OSSEOTOUCH team — clinical questions, configuration, Try Before You Buy (when available), financing.
Pick a date and time. We will contact you to discuss your workflow.
cs@osseotouch.com
Direct chat with our team.
U.S. ordersOrder through OSSEOTOUCH USA — fast nationwide delivery.
Medical device for professional use, restricted to qualified dental professionals according to the instructions for use. United States: registered with the U.S. FDA under Establishment Registration 3011922183 and product codes EIS · KDG · GEY (21 CFR 872.4565 / 878.4820). U.S. Agent: Thema USA, New York. ISO 13485. CE-marked. Manufactured by Meta Ergonomica S.r.l. (Italy). Clinical outcomes depend on case selection, surgical technique, and protocol.