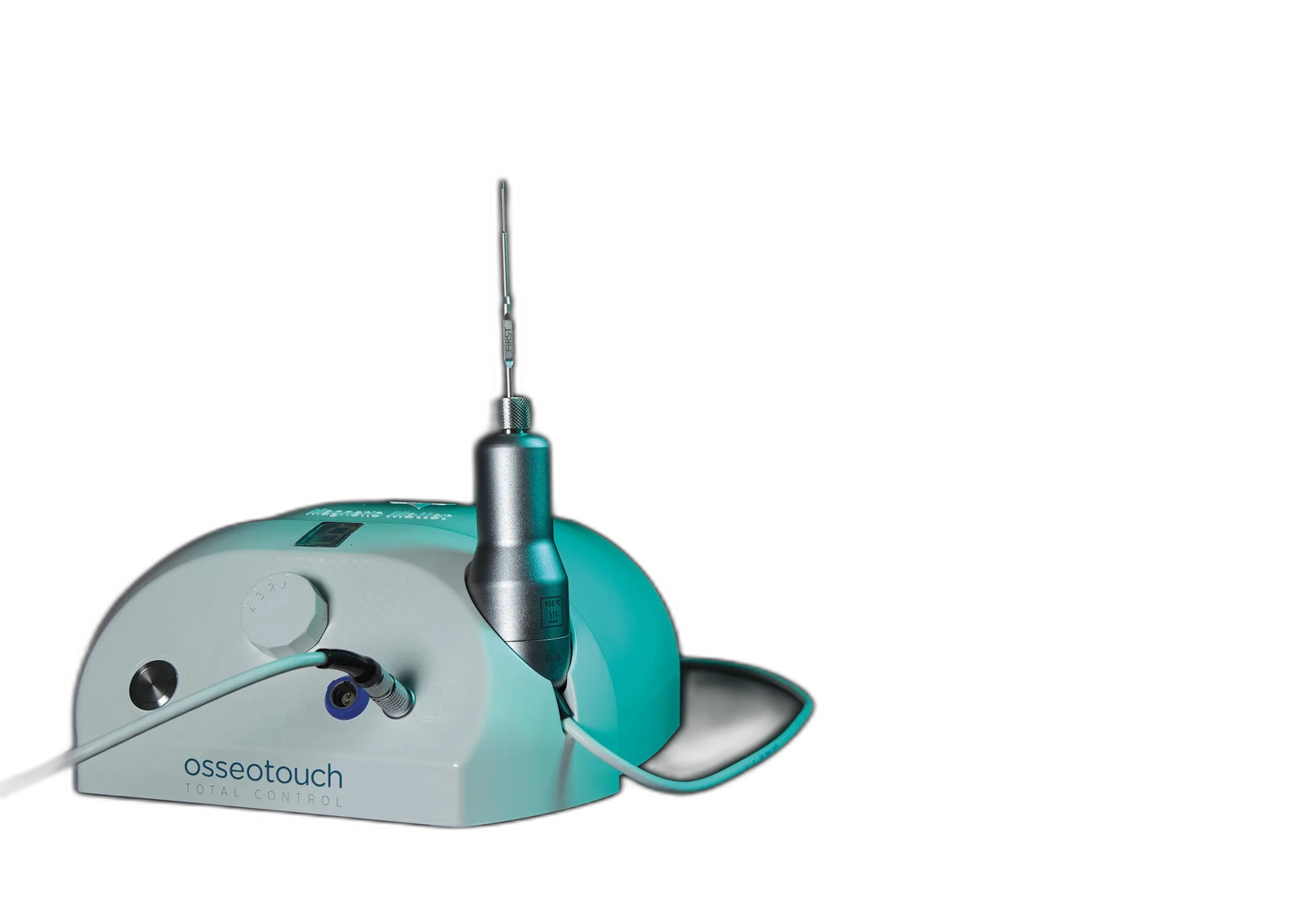
Magnetic Mallet
Calibrated force
Conserved bone
What changes when force is calibrated
- Bone preservation
- No rotary friction heat
- Surgeon-led control
Three published configurations
from entry-level to flagship
Three pre-composed bundles to start without choosing kit by kit. Each remains expandable at any time.
Infinity
Device + Essential Extraction Kit + Silver Osteotome Kit. 10 instruments. Atraumatic extractions, implant site preparation, free-hand crestal sinus lift.
Discover Infinity → PREMIUM STANDARDTotal Combo
Device + Essential + Black Ruby + Kit Elevate. 14 instruments. Next-generation rotary bur-free site preparation and millimetric-stop sinus lift. DLC coating.
Discover Total Combo → FLAGSHIPMaster
Device + Essential + BLEXO + Levacorone + Black Ruby + Kit Elevate. 26 instruments. The most complete published configuration.
Discover Master →
Latest-generation instrumentation
and dedicated kits
Magnetodynamic devices, atraumatic extractors, kits structured by clinical protocol. Twelve specialized kits, one handpiece.

BLEXO Extractors
Premium magnetodynamic extractors for the most challenging cases

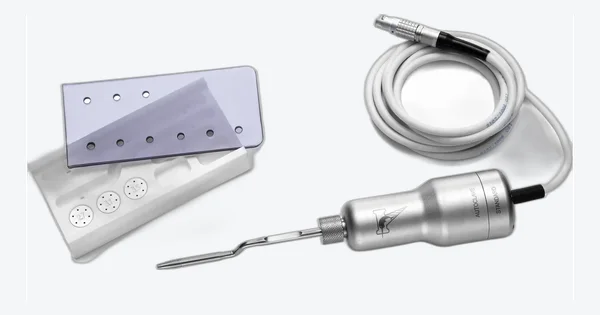
Essential Extraction Kit
Atraumatic everyday extractions, third molars included

Kit Elevate
Crestal sinus lift with millimetric stops, DLC coating

Black Ruby
Next-generation rotary bur-free implant site preparation

Ridge Expansion Kit
Horizontal split-crest expansion of atrophic ridges, no grafting

Kit Dynamic Guided
Magnetodynamic guided surgery — sleeve-compatible, no rotary heat

PT-1 Pterygoid
Pterygoid implant placement with axis control

Silver Osteotome Kit
Six progressive-diameter osteotomes for non-rotary osseodensification

Kit Easy In 6
20-degree-angle mandibular access osteotomies
Easy-Pin
Single-hand membrane fixation for GBR

First Lancet Osteotome
Pilot access and axis control on knife-edge ridges
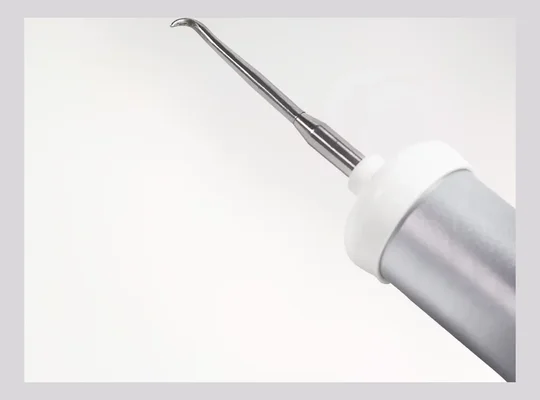
Levacorone (Crown Remover)
Low-force magnetodynamic crown and bridge removal
U.S. orders · ships nationwide
Order the Magnetic Mallet through OSSEOTOUCH — fast nationwide delivery, dedicated clinical support.
Learn magnetodynamic surgery
not just the instruments
A clinical discipline, not a transaction. Learn from the clinicians who developed magnetodynamic surgery — surrounded by peers who use it daily.
Live-surgery courses
Operate alongside the clinicians who developed magnetodynamic surgery. Hands-on, never theoretical.
02 Live · onlineLive webinars
Clinical cases and operating protocols, broadcast live. Q&A with specialists, attended from anywhere.
03 On demandClinical channel
Step-by-step tutorials, surgical recordings, and protocol walk-throughs from leading clinicians worldwide.
04 NetworkPeer community
An international WhatsApp community of clinicians who use the Magnetic Mallet daily. Trade cases, refine techniques, support difficult decisions.
Talk with the
OSSEOTOUCH team
Clinical questions, configuration, Try Before You Buy (when available), financing — handled by a specialist in a single dedicated session.
OSSEOTOUCH-branded medical devices are manufactured by Meta Ergonomica S.r.l. (Italy) and distributed under the OSSEOTOUCH brand by OSNRGY SRL. They are medical devices for professional use, restricted to qualified dental professionals according to the instructions for use. United States: Meta Ergonomica S.r.l. is registered and listed with the U.S. FDA under Establishment Registration 3011922183 and product codes EIS · KDG · GEY (21 CFR 872.4565 / 878.4820). U.S. Agent: Thema USA, New York. ISO 13485:2016 (Certificate ICIM-13485-050738-00 / IQNET IT-149738). CE-marked under EU MDR. Clinical outcomes depend on case selection, surgical technique, and protocol.